Ocean acidification
Ocean acidification: why does it happen and what are the consequences?
The carbon dioxide (CO2) emissions we release into the atmosphere not only worsen air quality, but also have an impact on the health of the oceans. Specifically, they cause a phenomenon known as acidification, which reduces the pH of the water, modifying its chemical composition and seriously affecting marine organisms. Solutions include reducing the use of fossil fuels in favour of renewable sources.

Climate change is a fact of life, and is proof of the impact of much human activity on the health of the planet. Although the commitment to sustainability and the environment has improved in recent years, the challenges are numerous and they include the pollution of the oceans -of particular concern is the presence of plastics- and their acidification. Below we will discuss this phenomenon, its causes, consequences and possible solutions.
What acidification is and why does it occur
First of all, we need to be clear about some concepts related to chemistry. Water can be of three types: acidic (if the pH is below seven), neutral (if the pH is seven) or alkaline (if the pH is above seven). That is, the lower the level of pH in water, which is measured on a scale from 0 to 14, the more acidic it is. The surface of the oceans, taken as a whole, has a pH ranging from 8.0 to 8.3. The marine organisms have developed and evolved in a environment have done with these pH values, so they are adapted to this ecosystem and any change may threaten their survival.
Ocean acidification occurs when, through a set of chemical reactions, seawater absorbs CO2. As noted in a 2019 report by the NOAA (National Oceanic and Atmospheric Administration), there is a positive side to this: were it not to occur, the concentration of CO2 in the atmosphere would be higher, which would further accelerate climate change. However, ocean acidification should not be seen as beneficial to marine life but rather as an undesirable consequence of excess CO₂ in the atmosphere. While the sea acts as a climate regulator, this environmental function comes at a high biological cost. Excessive carbon absorption disrupts processes essential for coral, molluscs and plankton. As a result, acidification represents an environmental problem that harms biodiversity, fisheries and the balance of marine ecosystems.
Despite this, acidification is not the only problem to be addressed. This phenomenon combines with other stressors such as ocean warming, deoxygenation, pollution and overfishing, exacerbating its effects.
How does ocean acidification affect people?
Ocean acidification is a serious phenomenon that directly harms people, especially those working in fisheries and aquaculture. By disrupting marine organisms, the balance of the food chain is upset, putting both food security and species conservation at risk. As a result, the availability of fish and shellfish decreases and their prices rise. This has a critical impact on coastal regions and all those whose income or daily diet depends entirely on the sea.
Este daño a los organismos del mar también se ve reflejado en el turismo, ya que la acidificación perjudica ecosistemas muy atractivos para visitantes, como los arrecifes de coral, que pierden color, biodiversidad y atractivo visual cuando se degradan. Con ello, actividades como el buceo, el snorkel y el turismo de playa se ven afectados, y puede golpear a negocios locales como hoteles, restaurantes y empresas de excursiones que dependen de estos paisajes marinos
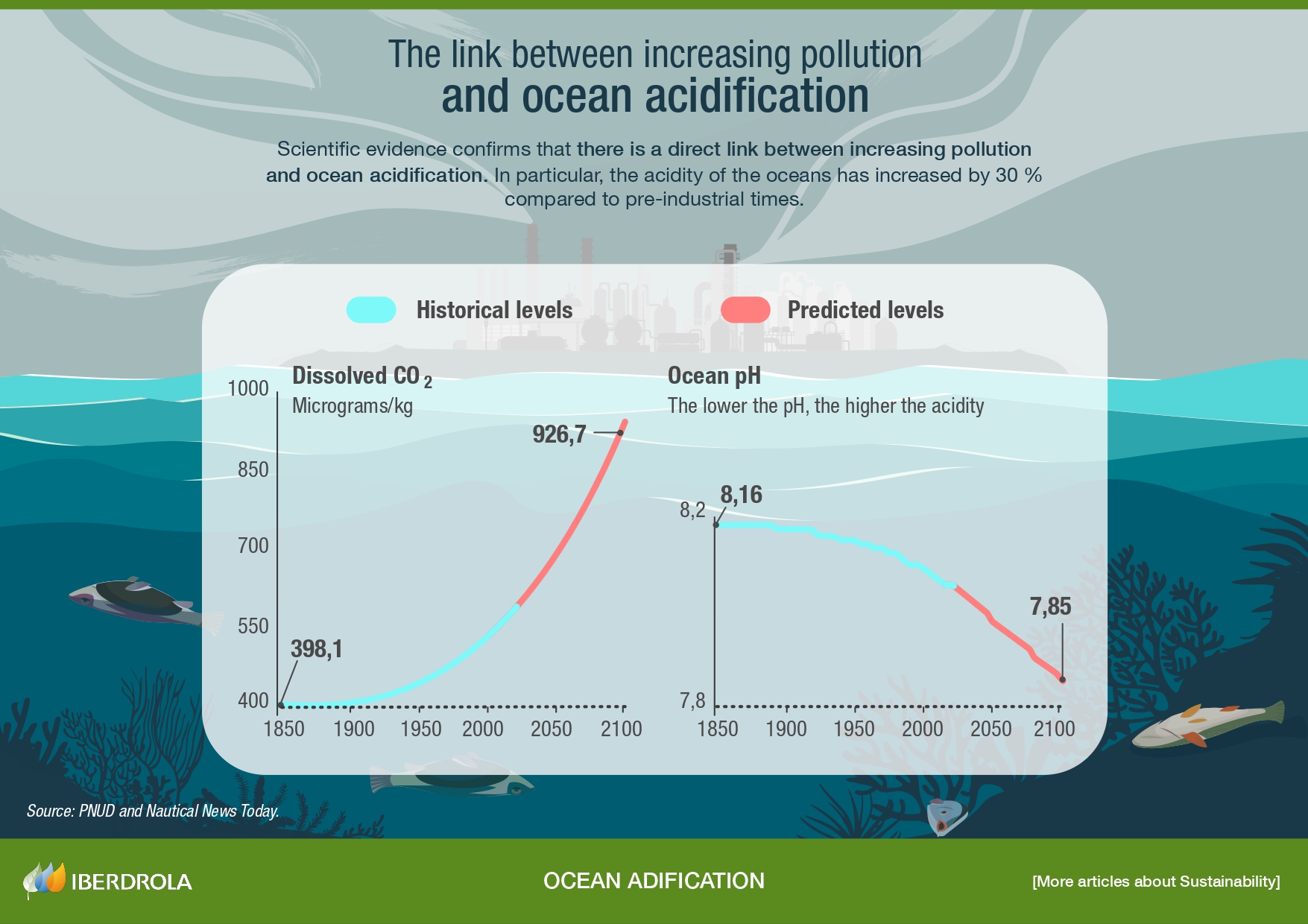
 SEE INFOGRAPHIC: The link between increasing pollution and ocean acidification [PDF]
SEE INFOGRAPHIC: The link between increasing pollution and ocean acidification [PDF]
Causes of ocean acidification
If we stick to the explanations given above, we could say that the main cause of ocean acidification is CO2 released into the atmosphere. Since the beginning of the Industrial Revolution, the concentration of this gas has increased severely due to the burning of fossil fuels used for industrial processes, power generation or transport, among other human activities.
In particular, CO2 levels in the atmosphere have increased from 250 to more than 400 parts per million (ppm) over the past centuries and, as a consequence, the pH of surface ocean water has decreased by 0.1. This may seem insignificant, but it represents a 30 % increase in ocean acidity compared to pre-industrial times. At current levels of fuel burning, pH is projected to decrease by a further 0.3 to 0.4 units by the end of this century, levels never seen in at least the last 25 million years and at a rate unprecedented in the entire history of the planet.
Consequences of ocean acidification
Who is affected by ocean acidification and how? According to Oceana, an NGO dedicated to marine conservation, we could categorise these consequences as follows:
Solutions to ocean acidification
Ocean acidification is a global problem, but there are clear strategies to tackle it. These actions fall mainly into two approaches: mitigation (tackling the root cause by reducing carbon emissions) and adaptation (minimising impacts and protecting affected communities and ecosystems).
Iberdrola and its commitment to caring for the oceans
For a company in the energy sector, the fight against ocean acidification is not a distant issue. Globally, the main solution lies in reducing CO2 emissions into the atmosphere, which are a direct consequence of human activities. To this end the Iberdrola Group has centred its strategy on processes such as electrification and decarbonisation, which are fundamental to minimising greenhouse gas emissions and achieving carbon neutrality. Through these measures we meet energy needs while at the same time contributing to the protection of marine ecosystems.
Specific measures for protecting the planet are integrated into our 2030 Biodiversity Plan through a conservation approach (avoid, reduce, restore and offset impacts). With this plan, we aim to avoid developing facilities in areas of high ecological value, reduce pollution and restore affected habitats. What’s more, we carry out quantifiable monitoring of our impact using measurement systems that assess the effects on species and ecosystems where our projects are located.
For example, at East Anglia One, where HVO30 fuel – composed entirely of vegetable oils – is used, emissions are reduced by 30% compared to traditional fuel. Similarly, we are studying the potential noise-related issues the plant may cause for animals, to ensure their welfare is not affected. Another significant project is Vineyard Wind 1, where eco-friendly cable protection mats are being deployed; these were designed to create more suitable habitats for the colonisation of benthic flora and fauna, that is, species living on the seabed.
These measures help build a better future and a cleaner, more sustainable energy model — a historic commitment that has made us leaders in electrical networks and renewables.












